THE TRUTH IS ALWAYS ON THE OTHER SIDE
Graphene-based gels can NOT find so-called "biomolecules" - the claim that this is the case, is also nonsense!!!
https://cdmf.org.br/wp-content/uploads/2020/07/Graphenic-Aerogels-Decorated-with-Ag-Nanoparticles-as-3D-SERS-Substrates-for-Biosensing.pdf
https://web.archive.org/web/20240405205704/https://cdmf.org.br/wp-content/uploads/2020/07/Graphenic-Aerogels-Decorated-with-Ag-Nanoparticles-as-3D-SERS-Substrates-for-Biosensing.pdf
Why is that? Because everything takes place in "experiments" in vitro with chemical substances/reagents and chemically-prepared cell lines/well plates/petri dish etc. using designed synthetic-chemically produced "oligo-nucleotide primers" - none of this has anything to do with the human/animal or plant organism in reality!!!
http://www.gene-quantification.org/castoldi-2-microrna-2008.pdf
https://web.archive.org/web/20240405211626/http://www.gene-quantification.org/castoldi-2-microrna-2008.pdf
So one must come to the only conclusion that graphene, graphene oxide and their derivatives are there solely to harm and kill people!
And it has been experimented with for many years⬇️ - nobody can say how many people have already been confronted with it without their knowledge, i.e. have been abused and had various clinical pictures/symptoms/manifestations as a result - from mild and serious health damage to long-term damage, cancer and death!!!
[1] K.S. Novoselov, A.K. Geim, S.V. Morozov, D. Jiang, Y. Zhang, S.V. Dubonos, I.V. Grigorieva, A.A. Firsov, Electric field effect in atomically thin carbon films, Science 306 (2004) 666–669.
[2] M.J. Allen, V.C. Tung, R.B. Kaner, Honeycomb carbon: a review of graphene, Chem. Rev. 110 (2010) 132–146.
[3] P.T. Yin, S. Shah, M. Chhowalla, K.B. Lee, Design, synthesis, and characterization of graphene-nanoparticle hybrid materials for bioapplications, Chem. Rev. 115 (2015) 2483–2531.
[4] K.S. Kim, Y. Zhao, H. Jang, S.Y. Lee, J.M. Kim, K.S. Kim, J.H. Ahn, P. Kim, J.Y. Choi, B.H. Hong, Large-scale pattern growth of graphene films for stretchable transparent electrodes, Nature 457 (2009) 706–710.
[5] R. Bogue, Graphene sensors: a review of recent developments, Sensor Rev. 34 (2014) 233–238.
[6] S. Ge, F. Lan, F. Yuc, J. Yu, Applications of graphene and related nanomaterials in analytical chemistry, New J. Chem. 39 (2015) 2380–2395.
[7] W.S. Hummers, R.E. Offeman, Preparation of graphitic oxide, J. Am. Chem. Soc. 80 (1958) 1339.
[8] C. Botas, P. Alvarez, P. Blanco, M. Granda, C. Blanco, R. Santamaría, L.J. Romasanta, R. Verdejo, M.A. Lopez-Manchado, R. Menendez, Graphene materials with different structures prepared from the same graphite by the Hummers and Brodie methods, Carbon 65 (2013) 156–164.
[9] D.C. Marcano, D.V. Kosynkin, J.M. Berlin, A. Sinitskii, Z. Sun, A. Slesarev, L.B. Alemany, W. Lu, J.M. Tour, Improved synthesis of graphene oxide, ACS Nano 4 (2010) 4806–4814.
[10] D. Li, M.B. Muller, S. Gilje, R.B. Kaner, G.G. Wallace, Processable aqueous dispersions of graphene nanosheets, Nat. Nanotechnol. 3 (2009) 101–105.
[11] Y. Zhu, S. Murali, W. Cai, X. Li, J.W. Suk, J.R. Potts, R.S. Ruoff, Graphene and graphene oxide: synthesis, properties, and applications, Adv. Mater. 22 (2010) 3906–3924.
[12] K.P. Loh, Q. Bao, G. Eda, M. Chhowalla, Graphene oxide as a chemically tunable platform for optical applications, Nat. Chem. 2 (2010) 1015–1024.
[13] A. Hunt, D.A. Dikin, E.Z. Kurmaev, T.D. Boyko, P. Bazylewski, G.S. Chang, A. Moewes, Epoxide speciation and functional group distribution in graphene oxide paper-like materials, Adv. Funct. Mater. 22 (2012) 3950–3957.
[14] W. Gao, L.B. Alemany, L. Ci, P.M. Ajayan, New insights into the structure and reduction of graphite oxide, Nat. Chem. 1 (2009) 403–408.
[15] Y. Liu, X. Dong, P. Chen, Biological and chemical sensors based on graphene materials, Chem. Soc. Rev. 41 (2012) 2283–2307.
[16] Y. Zhang, C. Wu, S. Guo, J. Zhang, Interactions of graphene and graphene oxide with proteins and peptides, Nanotechnol. Rev. 2 (2013) 27–45.
[17] Y. Wang, Z. Li, J. Wang, J. Li, Y. Lin, Graphene and graphene oxide: biofunctionalization and applications in biotechnology, Trends Biotechnol. 29 (2011) 205–212.
[18] C. Chung, Y.K. Kim, D. Shin, S.R. Ryoo, B.H. Hong, D.-H. Min, Biomedical applications of graphene and graphene oxide, Acc. Chem. Res. 46 (2013) 2211–2224.
[19] X. Huang, Z. Yin, S. Wu, X. Qi, Q. He, Q. Zhang, Q. Yan, F. Boey, H. Zhang, Graphenebased materials: synthesis, characterization, properties, and applications, Small 7 (2011) 1876–1902.
[20] S. Park, J. An, I. Jung, R.D. Piner, S.J. An, X. Li, A. Velamakanni, R.S. Ruoff, Colloidal suspensions of highly reduced graphene oxide in a wide variety of organic solvents, Nano Lett. 9 (2009) 1593–1597.
[21] Y. Si, E.T. Samulski, Synthesis of water soluble graphene, Nano Lett. 8 (2008) 1679–1682.
[22] J. Shen, Y. Hu, M. Shi, X. Lu, C. Qin, C. Li, M. Ye, Fast and facile preparation of graphene oxide and reduced graphene oxide nanoplatelets, Chem. Mater. 21 (2009) 3514–3520.
[23] S. Pei, H.-M. Cheng, The reduction of graphene oxide, Carbon 50 (2012) 3210–3228.
[24] S. Bai, X. Shen, Graphene-inorganic nanocomposites, RSC Adv. 2 (2012) 64–98.
[25] Y.K. Kim, M.H. Kim, D.-H. Min, Biocompatible reduced graphene oxide prepared by using dextran as a multifunctional reducing agent, Chem. Commun. 47 (2011) 3195–3197.
[26] S.K. Vashist, J.H.T. Luong, Recent advances in electrochemical biosensing schemes using graphene and graphene-based nanocomposites, Carbon 84 (2015) 519–550.
[27] J. Kim, L.J. Cote, F. Kim, W. Yuan, K.R. Shull, J. Huang, Graphene oxide sheets at interfaces, J. Am. Chem. Soc. 132 (2010) 8180–8186.
[28] J.J. Shao, W. Lv, Q.H. Yang, Self-assembly of graphene oxide at interfaces, Adv. Mater. 26 (2014) 5586–5612.
[29] R.S. Swathi, K.L. Sebastian, Resonance energy transfer from a dye molecule to graphene, J. Chem. Phys. 129 (2008) 054703.
[30] R.S. Swathi, K.L. Sebastian, Long range resonance energy transfer from a dye molecule to graphene has (distance)(−4) dependence, J. Chem. Phys. 130 (2009) 186010.
[31] J. Kim, L.J. Cote, F. Kim, J. Huang, Visualizing graphene based sheets by fluorescence quenching microscopy, J. Am. Chem. Soc. 132 (2010) 260–267.
[32] E. Morales-Narvaez, A. Merkoci, Graphene oxide as an optical biosensing platform, Adv. Mater. 24 (2012) 3298–3308.
[33] M. de Miguel, M. Alvaro, H. García, Graphene as a quencher of electronic excited states of photochemical probes, Langmuir 28 (2012) 2849–2857.
[34] E. Morales-Narváez, B. Pérez-López, L.B. Pires, A. Merkoçi, Simple Förster resonance energy transfer evidence for the ultrahigh quantum dot quenching efficiency by graphene oxide compared to other carbon structures, Carbon 50 (2012) 2987–2993.
[35] M. Wu, R. Kempaiah, P.J. Huang, V. Maheshwari, J. Liu, Adsorption and desorption of DNA on graphene oxide studied by fluorescently labeled oligonucleotides, Langmuir 27 (2011) 2731–2738.
[36] B. Liu, Z. Sun, X. Zhang, J. Liu, Mechanisms of DNA sensing on graphene oxide, Anal. Chem. 85 (2013) 7987–7993.
[37] H. Zhang, S. Jia, M. Lv, J. Shi, X. Zuo, S. Su, L. Wang, W. Huang, C. Fan, Q. Huang, Sizedependent programming of the dynamic range of graphene oxide-DNA interaction-based ion sensors, Anal. Chem. 86 (2014) 4047–4051.
[38] J. Lee, Y. Yim, S. Kim, M.-H. Choi, B.-S. Choi, Y. Lee, D.-H. Min, In-depth investigation of the interaction between DNA and nano-sized graphene oxide, Carbon 97 (2016) 92–98.
[39] X. Kong, Y. Huang, Applications of graphene in mass spectrometry, J. Nanosci. Nanotechnol. 14 (2014) 4719–4732.
[40] M. De, S.S. Chou, V.P. Dravid, Graphene oxide as an enzyme inhibitor: modulation of activity of α-chymotrypsin, J. Am. Chem. Soc. 33 (2011) 17524–17527.
[41] L.A.L. Tang, J. Wang, K.P. Loh, Graphene-based SELDI probe with ultrahigh extraction and sensitivity for DNA oligomer, J. Am. Chem. Soc. 132 (2010) 10976–10977.
[42] X. Ling, L. Xie, Y. Fang, H. Xu, H. Zhang, J. Kong, M.S. Dresselhaus, J. Zhang, Z. Liu, Can graphene be used as a substrate for Raman enhancement? Nano Lett. 10 (2009) 553–561.
[43] L. Xie, X. Ling, Y. Fang, J. Zhang, Z. Liu, Graphene as a substrate to suppress fluorescence in resonance raman spectroscopy, J. Am. Chem. Soc. 131 (2009) 9890–9891.
[44] W. Xu, N. Mao, J. Zhang, Graphene: a platform for surface-enhanced Raman spectroscopy, Small 9 (2013) 1206–1224.
[45] W. Xu, X. Ling, J. Xiao, M.S. Dresselhaus, J. Kong, H. Xu, Z. Liu, J. Zhang, Surface enhanced Raman spectroscopy on a flat graphene surface, Proc. Natl. Acad. Sci. 109 (2012) 9281–9286.
[46] B. Sharma, R.R. Frontiera, A.-I. Henry, E. Ringe, R.P.V. Duyne, SERS: materials, applications, and the future, Mater. Today 15 (2012) 16–25.
[47] C.H. Lu, H.H. Yang, C.L. Zhu, X. Chen, G.N. Chen, A graphene platform for sensing biomolecules, Angew. Chem. Int. Ed. 48 (2009) 4785–4787.
[48] S.J. He, B. Song, D. Li, C.F. Zhu, W.P. Qi, Y.Q. Wen, L.H. Wang, S.P. Song, H.P. Fang, C.H. Fan, A graphene nanoprobe for rapid, sensitive, and multicolor fluorescent DNA analysis, Adv. Funct. Mater. 20 (2010) 453–459.
[49] F. Li, Y. Huang, Q. Yang, Z. Zhong, D. Li, L. Wang, S. Song, C. Fan, A grapheneenhanced molecular beacon for homogeneous DNA detection, Nanoscale 2 (2010) 1021–1026.
[50] J.W. Yi, J. Park, N.J. Singh, I.J. Lee, K.S. Kim, B.H. Kim, Quencher-free molecular beacon: enhancement of the signal-to-background ratio with graphene oxide, Bioorg, Med. Chem. Lett. 21 (2011) 704–706.
[51] H. Dong, J. Zhang, H. Ju, H. Lu, S. Wang, S. Jin, K. Hao, H. Du, X. Zhang, Highly sensitive multiple microRNA detection based on fluorescence quenching of graphene oxide and isothermal strand-displacement polymerase reaction, Anal. Chem. 84 (2012) 4587–4593.
[52] L. Cao, L. Cheng, Z. Zhang, Y. Wang, X. Zhang, H. Chen, B. Liu, S. Zhang, J. Kong, Visual and high-throughput detection of cancer cells using a graphene oxide-based FRET aptasensing microfluidic chip, Lab Chip 12 (2012) 4864–4869.
[53] H. Chang, L. Tang, Y. Wang, J. Jiang, J. Li, Graphene fluorescence resonance energy transfer aptasensor for the thrombin detection, Anal. Chem. 82 (2010) 2341–2346.
[54] Y. Wen, F. Xing, S. He, S. Song, L. Wang, Y. Long, D. Li, C. Fan, A graphene-based fluorescent nanoprobe for silver(I) ions detection by using graphene oxide and a silver-specific oligonucleotide, Chem. Commun. 46 (2010) 2596–2598.
[55] Y. He, Z.G. Wang, H.W. Tang, D.W. Pang, Low background signal platform for the detection of ATP: when a molecular aptamer beacon meets graphene oxide, Biosens. Bioelectron. 29 (2011) 76–81.
[56] H. Pei, J. Li, M. Lv, J. Wang, J. Gao, J. Lu, Y. Li, Q. Huang, J. Hu, C. Fan, A graphenebased sensor array for high-precision and adaptive target identification with ensemble aptamers, J. Am. Chem. Soc. 134 (2012) 13843–13849.
[57] Z. Lu, L. Zhang, Y. Deng, S. Li, N. He, Graphene oxide for rapid microRNA detection, Nanoscale 4 (2012) 5840–5842.
[58] H.F. Dong, W. Gao, F. Yan, H. Ji, H. Ju, Fluorescence resonance energy transfer between quantum dots and graphene oxide for sensing biomolecules, Anal. Chem. 82 (2010) 5511–5517.
[59] C. Liu, Z. Wang, H. Jia, Z. Li, Efficient fluorescence resonance energy transfer between upconversion nanophosphors and graphene oxide: a highly sensitive biosensing platform, Chem. Commun. 47 (2011) 4661–4663.
[60] Y. Tao, Y. Lin, Z. Huang, J. Ren, X. Qu, DNA-templated silver nanoclustersgraphene oxide nanohybrid materials: a platform for label-free and sensitive fluorescence turn-on detection of multiple nucleic acid targets, Analyst 137 (2012) 2588–2592.
[61] X. Liu, F. Wang, R. Aizen, O. Yehezkeli, I. Willner, Graphene oxide/nucleic-acidstabilized silver nanoclusters: functional hybrid materials for optical aptamer sensing and multiplexed analysis of pathogenic DNAs, J. Am. Chem. Soc. 135 (2013) 11832–11839.
[62] S. Guo, D. Du, L. Tang, Y. Ning, Q. Yao, G.J. Zhang, PNA-assembled graphene oxide for sensitive and selective detection of DNA, Analyst 138 (2013) 3216–3220.
[63] J. Lee, I.S. Park, E. Jung, Y. Lee, D.-H. Min, Direct, sequence-specific detection of dsDNA based on peptide nucleic acid and graphene oxide without requiring denaturation, Biosens. Bioelectron. 62 (2014) 140–144.
[64] J. Lee, G. Park, D.-H. Min, A biosensor for the detection of single base mismatches in microRNA, Chem. Commun. 51 (2015) 14597–14600.
[65] J. Lee, I.S. Park, H. Kim, J.S. Woo, B.S. Choi, D.-H. Min, BSA as additive: a simple strategy for practical applications of PNA in bioanalysis, Biosens. Bioelectron. 69 (2015) 167–173.
[66] X.L. Dong, J. Cheng, J. Li, Y. Wang, Graphene as a novel matrix for the analysis of small molecules by MALDI-TOF MS, Anal. Chem. 82 (2010) 6208–6214.
[67] B. Gulbakan, E. Yasun, M. Shukoor, Z. Zhu, M. You, X. Tan, H. Sanchez, D.H. Powell, H. Dai, W. Tan, A dual platform for selective analyte enrichment and ionization in mass spectrometry using aptamer-conjugated graphene oxide, J. Am. Chem. Soc. 132 (2010) 17408–17410.
[68] Y. Liu, J. Liu, P. Yin, M. Gao, C. Deng, X. Zhang, High throughput identification of components from traditional Chinese medicine herbs by utilizing graphene or graphene oxide as MALDI-TOF-MS matrix, J. Mass Spectrom. 46 (2011) 804–815.
[69] C. Liu, M.W. Chien, C.Y. Su, H.Y. Chen, L.J. Li, C.C. Lai, Analysis of flavonoids by graphene-based surface-assisted laser desorption/ionization time-of-flight mass spectrometry, Analyst 137 (2012) 5809–5816.
[70] Q. Liu, M. Cheng, G. Jiang, Mildly oxidized graphene: facile synthesis, characterization, and application as a matrix in MALDI mass spectrometry, Chem. Eur. J. 19 (2013) 5561–5565.
[71] G. Cheng, Z.G. Wang, Y.L. Liu, J.L. Zhang, D.H. Sun, J.Z. Ni, A graphene-based multifunctional affinity probe for selective capture and sequential identification of different biomarkers from biosamples, Chem. Commun. 48 (2012) 10240–10242.
[72] H. Kawasaki, K. Nakai, R. Arakawa, E.K. Athanassiou, R.N. Grass, W.J. Stark, Wang, Functionalized graphene-coated cobalt nanoparticles for highly efficient surfaceassisted laser desorption/ionization mass spectrometry analysis, Anal. Chem. 84 (2012) 9268–9275.
[73] Y. Liang, X. He, L. Chen, Y. Zhang, Facile preparation of graphene/Fe3O4/TiO2 multifunctional composite for highly selective and sensitive enrichment of phosphopeptides, RSC Adv. 4 (2014) 18132–18135.
[74] R.C. Huang, W.J. Chiu, I. Po-Jung Lai, C.C. Huang, Multivalent aptamer/gold nanoparticle-modified graphene oxide for mass spectrometry-based tumor tissue imaging, Sci. Rep. 5 (2015) 10292.
[75] Y.K. Kim, H.K. Na, S.J. Kwack, S.R. Ryoo, Y. Lee, S. Hong, S. Hong, Y. Jeong, D.-H. Min, Synergistic effect of graphene oxide/MWCNT films in laser desorption/ionization mass spectrometry of small molecules and tissue imaging, ACS Nano 5 (2011) 4550–4561.
[76] Y.K. Kim, D.-H. Min, Fabrication of alternating multilayer films of graphene oxide and carbon nanotube and its application in mechanistic study of laser desorption/ ionization of small molecules, ACS Appl. Mater. Interfaces 4 (2012) 2088–2095.
[77] Y.K. Kim, D.-H. Min, Preparation of the hybrid film of poly(allylamine hydrochloride)-functionalized graphene oxide and gold nanoparticle and its application for laser-induced desorption/ionization of small molecules, Langmuir 28 (2012) 4453–4458.
[78] K. Qian, L. Zhou, J. Liu, J. Yang, H. Xu, M. Yu, A. Nouwens, J. Zou, M.J. Monteiro, C. Yu, Laser engineered graphene paper for mass spectrometry imaging, Sci. Rep. 3 (2013) 1415.
[79] M. Zhou, Y. Zhai, S. Dong, Electrochemical sensing and biosensing platform based on chemically reduced graphene oxide, Anal. Chem. 81 (2009) 5603–5613.
[80] L. Feng, L. Wu, J. Wang, J. Ren, D. Miyoshi, N. Sugimoto, X. Qu, Detection of a prognostic indicator in early stage cancer using functionalized graphene based peptide sensors, Adv. Mater. 23 (2012) 125–131.
[81] A. Bonanni, C.K. Chua, G. Zhao, Z. Sofer, M. Pumera, Inherently electroactive graphene oxide nanoplatelets as labels for single nucleotide polymorphism detection, ACS Nano 6 (2012) 8546–8551.
[82] L. He, Q. Wang, D. Mandler, M. Li, R. Boukherroub, S. Szunerits, Detection of folic acid protein in human serum using reduced graphene oxide electrodes modified by folic-acid, Biosens. Bioelectron. 75 (2016) 389–395.
[83] D. Choi, H. Jeong, K. Kim, Electrochemical thrombin detection based on the direct interaction of target proteins and graphene oxide as an indicator, Analyst 139 (2014) 1331–1333.
[84] J. Liu, N. Kong, A. Li, X. Luo, L. Cui, R. Wang, S. Feng, Graphene bridged enzyme electrodes for glucose biosensing application, Analyst 138 (2013) 2567–2575.
[85] Z.J. Wang, X. Zhou, J. Zhang, F. Boey, H. Zhang, Direct electrochemical reduction of single-layer graphene oxide and subsequent functionalization with glucose oxidase, J. Phys. Chem. C 113 (2009) 14071–14075.
[86] S. Srivastava, S. Abraham, C. Singh, M.A. Ali, A. Srivastava, G. Sumanaa, B.D. Malhotra, Protein conjugated carboxylated gold@reduced graphene oxide for aflatoxin B1 detection, RSC Adv. 5 (2015) 5406–5414.
[87] J. Li, S. Guo, Y. Zhai, E. Wang, High-sensitivity determination of lead and cadmium based on the nafion-graphene composite film, Anal. Chim. Acta 649 (2009) 196–201.
[88] J. Gong, T. Zhou, D. Song, L. Zhang, Monodispersed Au nanoparticles decorated graphene as an enhanced sensing platform for ultrasensitive stripping voltammetric detection of mercury(II), Sensors Actuators B 150 (2010) 491–497.
[89] C.X. Guo, X.T. Zheng, Z.S. Lu, X.W. Lou, C.M. Li, Biointerface by cell growth on layered graphene-artificial peroxidase–protein nanostructure for in situ quantitative molecular detection, Adv. Mater. 22 (2010) 5164–5167.
[90] L. Feng, Y. Chen, J. Ren, X. Qu, A graphene functionalized electrochemical aptasensor for selective label-free detection of cancer cells, Biomaterials 32 (2011) 2930–2937.
[91] S.M. Khoshfetrat, M.A. Mehrgardi, Amplified electrochemical genotyping of singlenucleotide polymorphisms using a graphene–gold nanoparticles modified glassy carbon platform, RSC Adv. 5 (2015) 29285–29293.
[92] Z.J. Wang, J. Zhang, P. Chen, X. Zhou, Y. Yang, S. Wu, L. Niu, Y. Han, L. Wang, P. Chen, F. Boey, Q. Zhang, B. Liedberg, H. Zhang, Label-free, electrochemical detection of methicillin-resistant Staphylococcus aureus DNA with reduced graphene oxidemodified electrodes, Biosens. Bioelectron. 26 (2011) 3881–3886.
[93] Y. Du, S. Guo, S. Dong, E. Wang, An integrated sensing system for detection of DNA using new parallel-motif DNA triplex system and graphene-mesoporous silicagold nanoparticle hybrids, Biomaterials 32 (2011) 8584–8592.
[94] S. Roy, N. Soin, R. Bajpai, D.S. Misra, J.A. McLaughlinb, S.S. Roy, Graphene oxide for electrochemical sensing applications, J. Mater. Chem. 21 (2011) 14725–14731.
[95] A.H. Loo, A. Bonanni, M. Pumera, Impedimetric thrombin aptasensor based on chemically modified graphenes, Nanoscale 4 (2012) 143–147.
[96] W. Ren, Y. Fang, E. Wang, A binary functional substrate for enrichment and ultrasensitive SERS spectroscopic detection of folic acid using graphene oxide/Ag nanoparticle hybrids, ACS Nano 5 (2011) 6425–6433.
[97] Z. Fan, R. Kanchanapally, P.C. Ray, Hybrid graphene oxide based ultrasensitive SERS probe for label-free biosensing, J. Phys. Chem. Lett. 4 (2013) 3813–3818.
[98] Y. Xie, Y. Li, L. Niu, H. Wang, H. Qian, W. Yao, A novel surface-enhanced Raman scattering sensor to detect prohibited colorants in food by graphene/silver nanocomposite, Talanta 100 (2012) 32–37.
[99] Y. Zhang, S. Liu, L. Wang, X. Qin, J. Tian, W. Lu, G. Changa, X. Sun, One-pot green synthesis of Ag nanoparticles-graphene nanocomposites and their applications in SERS, H2O2, and glucose sensing, RSC Adv. 2 (2012) 538–545.
[100] Y. Li, L.L. Qu, D.W. Li, Q.X. Song, F. Fathi, Y.T. Long, Rapid and sensitive in-situ detection of polar antibiotics in water using a disposable Ag–graphene sensor based on electrophoretic preconcentration and surface-enhanced Raman spectroscopy, Biosen. Bioelectron. 43 (2013) 94–100.
[101] T.-W. Lin, H.-Y. Wu, T.-T. Tasi, Y.-H. Laia, H.-H. Shen, Surface-enhanced Raman spectroscopy for DNA detection by the self-assembly of Ag nanoparticles onto Ag nanoparticle-graphene oxide nanocomposites, Phys. Chem. Chem. Phys. 17 (2015) 18443–18448.
[102] C. Hu, Y. Liu, J. Qin, G. Nie, B. Lei, Y. Xiao, M. Zheng, J. Rong, Fabrication of reduced graphene oxide and sliver nanoparticle hybrids for Raman detection of absorbed folic acid: a potential cancer diagnostic probe, ACS Appl. Mater. Interfaces 5 (2013) 4760–4768.
[103] Z. Zhang, Q. Liu, D. Gao, D. Luo, Y. Niu, J. Yang, Y. Li, Graphene Oxide as a Multifunctional Platform for Raman and Fluorescence Imaging of Cells. Small 11 (2015) 3000–3005.
[104] Y. K. Kim, S. W. Han, D.-H. Min, Graphene oxide sheath on Ag nanoparticle/ graphene hybrid films as an antioxidative coating and enhancer of surfaceenhanced Raman scattering, ACS Appl. Mater. Interfaces 4 (1021) 6545–6551.
[105] N. Gao, T. Yang, T. Liu, Y. Zoua, J. Jiang, Graphene oxide wrapped individual silver nanocomposites with improved stability for surface-enhanced Raman scattering, RSC Adv. 5 (2015) 55801–55807.
[106] L. Cui, X. Lin, N. Lin, Y. Song, Z. Zhu, X. Chen, C.J. Yang, Graphene oxide-protected DNA probes for multiplex microRNA analysis in complex biological samples based on a cyclic enzymatic amplification method, Chem. Commun. 48 (2012) 194–196.
[107] Y. Pu, Z. Zhu, D. Han, H. Liu, J. Liu, J. Liao, K. Zhang, W. Tan, Insulin-binding aptamerconjugated graphene oxide for insulin detection, Analyst 136 (2011) 4138–4140.
[108] X. Liu, R. Aizen, R. Freeman, O. Yehezkeli, I. Willner, Multiplexed aptasensors and amplified DNA sensors using functionalized graphene oxide: application for logic gate operations, ACS Nano 6 (2012) 3553–3563.
[109] R.C. Huang, W.J. Chiu, Y.J. Li, C.C. Huang, Detection of microRNA in tumor cells using exonuclease III and graphene oxide-regulated signal amplification, ACS Appl. Mater. Interfaces 6 (2014) 21780–21787.
[110] Y. Liu, M. Luo, X. Xiang, C. Chen, X. Ji, L. Chen, Z. He, A graphene oxide and exonuclease-aided amplification immuno-sensor for antigen detection, Chem. Commun. 50 (2014) 2679–2681.
[111] C.H. Lu, J. Li, M.H. Lin, Y.W. Wang, H.H. Yang, X. Chen, G.N. Chen, Amplified aptamer-based assay through catalytic recycling of the analyte, Angew. Chem. Int. Ed. 49 (2010) 8454–8457.
[112] X. Li, X. Ding, J. Fan, Nicking endonuclease-assisted signal amplification of a split molecular aptamer beacon for biomolecule detection using graphene oxide as a sensing platform, Analyst 140 (2015) 7918–7925.
[113] Z. Li, W. Zhu, J. Zhang, J. Jiang, G. Shen, R. Yu, A label-free amplified fluorescence DNA detection based on isothermal circular strand-displacement polymerization reaction and graphene oxide, Analyst 138 (2013) 3616–3620.
[114] K. Hu, J. Liu, J. Chen, Y. Huang, S. Zhao, J. Tian, G. Zhang, An amplified graphene oxide-based fluorescence aptasensor based on target-triggered aptamer hairpin switch and strand-displacement polymerization recycling forbioassays, Biosens. Bioelectron. 42 (2013) 598–602.
[115] H. Liu, L. Li, Q. Wang, L. Duan, B. Tang, Graphene fluorescence switch-based cooperative amplification: a sensitive and accurate method to detection microRNA, Anal. Chem. 86 (2014) 5487–5493.
[116] T. Wang, Z. Zhang, Y. Lib, G. Xie, Amplified electrochemical detection of mecA gene in methicillin-resistant Staphylococcus aureus based on target recycling amplification and isothermal strand-displacement polymerization reaction, Sensors Actuators B Chem. 221 (2015) 148–154.
[117] M. Liu, J. Song, S. Shuang, C. Dong, J.D. Brennan, Y. Li, A graphene-based biosensing platform based on the release of DNA probes and rolling circle amplification, ACS Nano 8 (2014) 5564–5573.
[118] X. Zhao, R.M. Kong, X.B. Zhang, H.M. Meng, W.N. Liu, W. Tan, G.L. Shen, R.Q. Yu, Graphene-DNAzyme based biosensor for amplified fluorescence “turn-on” detection of Pb2+ with a high selectivity, Anal. Chem. 83 (2011) 5062–5066.
[119] M. Liu, H. Zhao, S. Chen, H. Yu, Y. Zhang, X. Quan, A “turn-on” fluorescent copper biosensor based on DNA cleavage-dependent graphene-quenched DNAzyme, Biosens. Bioelectron. 26 (2011) 4111–4116.
[120] L. Yang, C. Liu, W. Ren, Z. Li, Graphene surface-anchored fluorescence sensor for sensitive detection of microRNA coupled with enzyme-free signal amplification of hybridization chain reaction, ACS Appl. Mater. Interfaces 4 (2012) 6450–6453.
[121] Z. Zhang, Y. Liu, X. Ji, X. Xiang, Z. He, A graphene oxide-based enzyme-free signal amplification platform for homogeneous DNA detection, Analyst 139 (2014) 4806–4809.
[122] J. Ge, Z.M. Huang, Q. Xi, R.Q. Yu, J.H. Jiang, X. Chu, A novel graphene oxide based fluorescent nanosensing strategy with hybridization chain reaction signal amplification for highly sensitive biothiol detection, Chem. Commun. 50 (2014) 11879–11882.
[123] L. Li, J. Feng, H. Liu, Q. Li, L. Tong, B. Tang, Two-color imaging of microRNA with enzyme-free signal amplification via hybridization chain reactions in living cells, Chem. Sci. 7 (2016) 1940–1945.
[124] H. Jang, Y.K. Kim, H.M. Kwon, W.S. Yeo, D.E. Kim, D.-H. Min, A graphene-based platform for the assay of duplex-DNA unwinding by helicase, Angew. Chem. Int. Ed. 49 (2010) 5703–5707.
[125] L. Lin, Y. Liu, X. Zhao, J. Li, Sensitive and rapid screening of T4 polynucleotide kinase activity and inhibition based on coupled exonuclease reaction and graphene oxide platform, Anal. Chem. 83 (2011) 8396–8402.
[126] J. Lee, Y.K. Kim, D.-H. Min, A new assay for endonuclease/methyltransferase activities based on graphene oxide, Anal. Chem. 83 (2011) 8906–8912.
[127] J.S. Park, A. Baek, I.S. Park, B.H. Jun, D.E. Kim, A graphene oxide-based platform for the assay of RNA synthesis by RNA polymerase using a fluorescent peptide nucleic acid probe, Chem. Commun. 49 (2013) 9203–9205.
[128] J. Lee, D.-H. Min, A simple fluorometric assay for DNA exonuclease activity based on graphene oxide, Analyst 137 (2012) 2024–2026.
[129] D.M. Zhou, Q. Xi, M.F. Liang, C.H. Chen, L.J. Tang, J.H. Jiang, Graphene oxide-hairpin probe nanocomposite as a homogeneous assay platform for DNA base excision repair screening, Biosens, Bioelectron 41 (2013) 359–365.
[130] L. Peng, Z. Zhu, Y. Chen, D. Han, W. Tan, An exonuclease III and graphene oxideaided assay for DNA detection, Biosens. Bioelectron. 35 (2012) 475–478.
[131] M. Zhang, B.C. Yin, X.F. Wang, B.C. Ye, Interaction of peptides with graphene oxide and its application for real-time monitoring of protease activity, Chem. Commun. 47 (2011) 2399–2401.
[132] J. Zhou, X. Xu, W. Liu, X. Liu, Z. Nie, M. Qing, L. Nie, S. Yao, Graphene oxide-peptide nanocomplex as a versatile fluorescence probe of protein kinase activity based on phosphorylation protection against carboxypeptidase digestion, Anal. Chem. 85 (2013) 5746–5754.
[133] F. Wang, C. Liu, Y. Fan, Y. Wang, Z. Li, Robust detection of tyrosine phosphatase activity by coupling chymotrypsin-assisted selective peptide cleavage and a graphene oxide-based fluorescent platform, Chem. Commun. 50 (2014) 8161–8163.
[134] J. Li, C.H. Lu, Q.H. Yao, X.L. Zhang, J.J. Liu, H.H. Yang, G.N. Chen, A graphene oxide platform for energy transfer-based detection of protease activity, Biosens. Bioelectron. 26 (2011) 3894–3899.
[135] E. Song, D. Cheng, Y. Song, M. Jiang, J. Yu, Y. Wang, A graphene oxide-based FRET sensor for rapid and sensitive detection of matrix metalloproteinase 2 in human serum sample, Biosens. Bioelectron. 47 (2013) 445–450.
[136] Y. Liu, Y. Li, J. Liu, C. Deng, X. Zhang, High throughput enzyme inhibitor screening by functionalized magnetic carbonaceous microspheres and graphene oxidebased MALDI-TOF-MS, J. Am. Soc. Mass. Spectr. 22 (2011) 2188–2198.
[137] J. Lee, Y.K. Kim, D.-H. Min, Laser desorption/ionization mass spectrometric assay for phospholipase activity based on graphene oxide/carbon nanotube double-layer films, J. Am. Chem. Soc. 132 (2010) 14714–14717.
[138] C.S. Shan, H. Yang, J. Song, D. Han, A. Ivaska, L. Niu, Direct electrochemistry of glucose oxidase and biosensing for glucose based on graphene, Anal. Chem. 81 (2009) 2378–2382.
[139] W. Li, P. Wu, H. Zhang, C. Cai, Signal amplification of graphene oxide combining with restriction endonuclease for site-specific determination of DNA methylation and assay of methyltransferase activity, Anal. Chem. 84 (2012) 7583–7590.
[140] L. Wu, J. Wang, L. Feng, J. Ren, W. Wei, X. Qu, Label-free ultrasensitive detection of human telomerase activity using porphyrin-functionalized graphene and electrochemiluminescence technique, Adv. Mater. 24 (2012) 2447–2452.
[141] L. Zhang, J. Liang, Y. Huang, Y. Ma, Y. Wang, Y. Chen, Size-controlled synthesis of graphene oxide sheets on a large scale using chemical exfoliation, Carbon 47 (2009) 3365–3368.
[142] J. Luo, L.J. Cote, V.C. Tung, A.T.L. Tan, P.E. Goins, J. Wu, J. Huang, Graphene oxide nanocolloids, J. Am. Chem. Soc. 132 (2010) 17667–17669.
[143] X. Wang, H. Bai, G. Shi, Size fractionation of graphene oxide sheets by pH-assisted selective sedimentation, J. Am. Chem. Soc. 133 (2011) 6338–6342.
[144] Y. Wang, Z. Li, D. Hu, C.T. Lin, J. Li, Y. Lin, Aptamer/graphene oxide nanocomplex for in situ molecular probing in living cells, J. Am. Chem. Soc. 132 (2010) 9274–9276.
[145] H.B. Wang, Q. Zhang, X. Chu, T. Chen, J. Ge, R. Yu, Graphene oxide-peptide conjugate as an intracellular protease sensor for caspase-3 activation imaging in live cells, Angew. Chem. Int. Ed. 50 (2011) 7065–7069.
[146] S.R. Ryoo, J. Lee, J. Yeo, H.K. Na, Y.K. Kim, H. Jang, J.H. Lee, S.W. Han, Y. Lee, V.N. Kim, D.-H. Min, Quantitative and multiplexed microRNA sensing in living cells based on peptide nucleic acid and nano graphene oxide (PANGO), ACS Nano 7 (2013) 5882–5891.
[147] S. Kim, S.R. Ryoo, H.K. Na, Y.K. Kim, B.S. Choi, Y. Lee, D.E. Kim, D.-H. Min, Deoxyribozyme-loaded nano-graphene oxide for simultaneous sensing and silencing of the hepatitis C virus gene in liver cells, Chem. Commun. 49 (2013) 8241–8243.
[148] Y. Wang, Z. Li, T.J. Weber, D. Hu, C.T. Lin, J. Li, Y. Lin, In situ live cell sensing of multiple nucleotides exploiting DNA/RNA aptamers and graphene oxide nanosheets, Anal. Chem. 85 (2013) 6775–6782.
[149] Y. Wang, L. Tang, Z. Li, Y. Lin, J. Li, In situ simultaneous monitoring of ATP and GTP using a graphene oxide nanosheet–based sensing platform in living cells, Nat. Protoc. 9 (2014) 1944–1955.
[150] Z. Liu, C. Hu, S. Li, W. Zhang, Z. Guo, Rapid intracellular growth of gold nanostructures assisted by functionalized graphene oxide and its application for surfaceenhanced Raman spectroscopy, Anal. Chem. 84 (2012) 10338–10344.
[151] Q.H. Liu, L. Wei, J. Wang, F. Peng, D. Luo, R. Cui, Y. Niu, X. Qin, Y. Liu, H. Sun, J. Yang, Y. Li, Cell imaging by graphene oxide based on surface enhanced Raman scattering, Nanoscale 4 (2012) 7084–7089.
[152] H. Jang, S.R. Ryoo, Y.K. Kim, S. Yoon, H. Kim, S.W. Han, B.S. Choi, D.E. Kim, D.-H. Min, Discovery of hepatitis C virus NS3 helicase inhibitors by a multiplexed, highthroughput helicase activity assay based on graphene oxide, Angew. Chem. Int. Ed. 52 (2013) 2340–2344.
https://www.dropbox.com/s/xcfp8arvikjgi21/2-%20GRAPHENE%20OXIDE%20TOXICITY%20REPORT.pdf?dl=0
https://www.sciencedirect.com/science/article/pii/S2590238521000679
https://www.cell.com/matter/pdf/S2590-2385(21)00067-9.pdf
https://web.archive.org/web/20240405230146/https://www.cell.com/matter/pdf/S2590-2385(21)00067-9.pdf
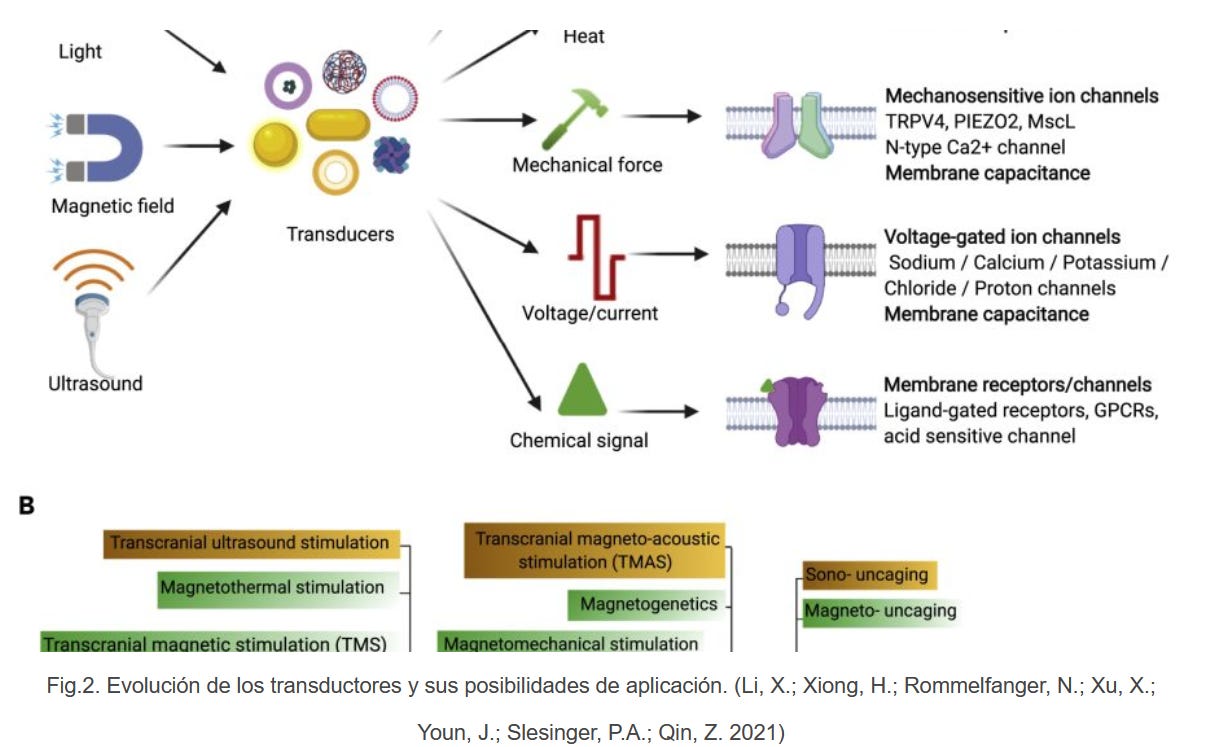
https://web.archive.org/web/20220208201159/https://corona2inspect.net/2021/07/19/el-oxido-de-grafeno-y-la-estimulacion-cerebral-por-ondas-electromagneticas-em-capaces-de-inferir-en-la-mente-humana/
Graphene oxide and brain stimulation by EM electromagnetic waves able to infer the human mind!
With my thoughts and my heart with you all - always!!!♥️♥️♥️