Reference
Li, J.M .; Chang, P.H.; Li, L .; Teo, C.J.; Khoo, B.C.; Duan, H.; Mai, V.C. (2018). Application of graphene oxide in Jet A-1 in air to improve the combustion process = Application of Graphene Oxide in Jet A-1 in Air to Enhance Combustion Process. In 2018 AIAA Aerospace Sciences Meeting, pp. 133-138. https://doi.org/10.2514/6.2018-0133
Introduction
From the article on the CO2 adsorption and absorption capacity and specifically the detail of the presence of graphene oxide in aerosols in the atmosphere, as a result of the incomplete combustion of jet aircraft (Pöschl, U. 2005), the investigation has been opened to the area of aviation fuels. This is because the presence of graphene oxide in aerosols in the atmosphere can only be due to the aerial vector or due to contamination caused by the fuels used by jet engines, or by direct effect or practice of intentional fumigation at high level. It must be considered that fumigation at a very low level, carried out by fumigation planes for the treatment of agricultural fields, should not influence the upper layers of the atmosphere, where the study of (Pöschl, U. 2005).
On the other hand, the subject of the analyzed article "Application of graphene oxide in Jet A-1 in air to improve the combustion process", transfers some concepts that must be clarified. Firstly, the types of air fuel and in particular Jet A-1 fuel. Usually air fuel, also known as kerosene, is an oil derivative that has great calorific value, lubricating capacity, easily injectable in jet engine turbines and a flammability point around 38ºC. Aviation fuels are classified according to civil or military use. Civil fuel has three variants, namely Jet-A, Jet-A1, and Jet-B. Jet-A1 fuel has a slightly lower freezing point (-47ºC) and a slight difference in energy density compared to Jet-A fuel. Jet-B fuel includes additives that allow it to operate at colder temperatures,since its freezing point is at -60ºC. In the military domain, there are JP1-10 fuels whose additives have been changing in successive versions to improve engine performance, reduce waste and facilitate subsequent maintenance of turbines.
Acts
The study of (Li, J.M .; Chang, P.H.; Li, L .; Teo, C.J.; Khoo, B.C.; Duan, H.; Mai, V.C. 2018) clearly presents the application of graphene oxide "GO" as an additive to Jet-A1 aviation fuel, as indicated as "investigates the feasibility of applying graphene oxide (GO) nanohojas to Jet A-1 to improve its combustion performance in the air, such as deflagration ignition delay, flame velocity, and shock-induced reaction flow".
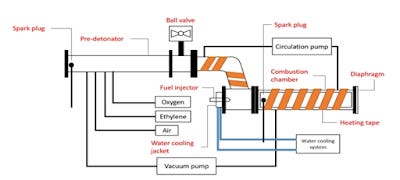
Fig. 1. Diagram of the combustion experiment at high speeds of the GO-Jet-A1 fuel. (Li, J.M .; Chang, P.H.; Li, L .; Teo, C.J.; Khoo, B.C.; Duan, H.; Mai, V.C. 2018)
In the words of the authors "the results indicate that a longer vaporization duration of the Jet A-1 or a higher chamber temperature results in smaller and more densely wrinkled GO particles that retain a high surface area as a potential microcatalyst to improve combustion reactions". This means that during the injection process of Jet-A1 fuel in the combustion chamber of the jet engine, it converts graphene oxide into particles that better adsorb fuel, which favors the speed with which the ignition reaction is triggered. This is stated as follows "the initial deflagration test of GO-Jet-A1 mixtures shows that the addition of GO nanohojas accelerates the initial linear combustion rate and reduces ignition delay times". Experiment data shows that "for 17.9% Jet A-1 fuel in air, the addition of GO in proportion to (2mg / ml) increases the initial linear combustion rate of 4.52m / sa5.15m / s (13.8%) and reduces ignition delay times from 8,195 ms to 3,045ms (62.8%)". An extraordinary detail is that the GO-Jet-A1 graphene oxide fuel "has photoignition properties and lower minimum ignition energy". This is very relevant, since it is easier to detonate even with a pulse of energy emitted from a xenon flash camera, making it an appropriate resource for solid fuel in rockets. In fact, the study refers directly to this phenomenon as follows "ethanol-soaked GO foam has shown that GO foam is capable of igniting ethanol vapor when lit with a xenon flash lamp"
Another interesting detail is the "possible inclusion of GO and / or metal nanoparticles (for example, Fe, Au, Pt, Cu ...) in aviation fuels, that would pave another way to improve the transition from deflagration to detonation in a pulse engine". This statement is relevant because it means that Fe3O4 nanoparticles with graphene oxide, already mentioned in the CO2 adsorption article, are compatible with mixing in aviation fuels.
The authors conclude that the "GO energy density and its high reactivity associated with metal nanoparticles, make them unique combustible additives in propellant formulations, for significantly higher and faster energy release". In fact they are not the only ones to study it.
Other studies
The work of (Askari, S.; Lotfi, R.; Rashidi, A.M .; Koolivand, H.; Koolivand-Salooki, M. 2016) also addresses the study of graphene oxide in the form of nanofluid, combined with kerosene, to determine the rheological, thermophysical and energy conservation properties. Interestingly, graphene oxide nanoparticles combined with Fe3O4, coated by oleic acid and combined with kerosene were used to carry out the experiment. The nanofluid obtained increased its viscosity, remaining more than five months without sedimentation. The maximum improvement in heat transfer was 66% with a weight increase of only 0.3% of the fuel.
Research on kerosene and graphene oxide additives continued in the work of ( Askari, S.; Rashidi, A.; Koolivand, H. 2019 ) to determine the behavior of a fuel combined with MWCNT ( Multi-Walled Carbon Nanotube ) or what is the same multi-walled carbon nanotubes, which are essentially concentric graphene oxide nanotubes. Among the results, they observed an improvement in convection heat transfer of 40.26%, confirming the "ultra-stability" of the compound.
Fig. 2. MWCNT carbon nanotubes in Table c ) obtained from the work of ( Rafique, I.; Kausar, A.; Anwar, Z.; Muhammad, B. 2016 )
Another study related to MWCNT carbon nanotubes is (Khaled, M. 2015) that guides its application to the catalytic function of elimination of thiophene and dibenzothiophene in diesel fuel. Among their most relevant results, they obtain an elimination of 68.8% of these compounds.
Research of (Agarwal, D.K .; Vaidyanathan, A.; Kumar, S.S. 2016) also addresses the use of kerosene-graphene nanofluids. In this case, the application of the fuel would be framed in the rocket engines. In addition to corroborating an improvement in combustion performance, an ideal property for these applications was found which is "regenerative cooling of semi-cryogenic rocket engines".
Work has also been done on graphene oxide additives for industrial fuels, in order to reduce sulfur oxide emissions, as they explain (Betiha, M.A.; Rabie, A.M .; Ahmed, H.S.; Abdelrahman, A.A.; El-Shahat, M.F. 2018). As indicated "The oxidative desulfurization process using graphene oxide has attracted significant interest in removing sulfur from fuels". Also, researchers add "The combustion of fossil fuels containing sulfur compounds emits some of the sulfur oxides that are considered a harmful influence on human health and the surrounding environment, as well as the economy". This statement is very surprising, considering the pernicious effects of graphene oxide, who do not seem to know the authors. In conclusion of their study they indicate that graphene oxide "GO remains an ideal type of catalyst for pure fuel in the near future due to its eligible physicochemical characteristics".
Another example of air fuel is (Dai, Y.; Nie, G.; Gong, S.; Wang, L .; Pan, L .; Fang, Y.; Zou, J.J. 2020) in which a low freezing point, high density, high calorific value and thermal stability of combustion are sought. To do this, they take biomass-based biofuels (derived from cellulose) combined with graphene oxide, so that the emulsification of the added reagents (cyclopentanol, methylcyclopentane and sulfuric acid). This achieved a mixture in a single tank, with a carbon yield of 83.2% and an improved performance compared to aviation fuel of 97.3%.
Another example of graphene oxide in aviation fuel is the one proposed by (Feng, M .; Jiang, X.Z.; Mao, Q.; Luo, K.H.; Hellier, P. 2019) to improve oxidation of the JP-10 propellant with functionalized graphene sheets. The JP-10 is a fuel used in missiles, military jet engines, ramjets and scramjets, whose characteristics are a high energy density, heat, thermal stability and low freezing point. The researchers corroborate that "graphene nanoplasts perform outstandingly by producing a more than 7% increase in combustion rate with just a particle load of 0.1%". In the discussion of their results they refer that "both pyrolysis and oxidation of JP-10 advance and improve in the presence of FGS (Functionalized graphene sheets), leading to earlier decomposition of JP-10 at a lower temperature and faster reaction rate". They conclude by stating that "This research lays the scientific foundation for the potential use of FGS as a promising catalyst for JP-10 fuel systems.". These statements are also recognized by the study of (Yadav, A.K .; Nandakumar, K .; Srivastava, A.; Chowdhury, A. 2019).
Powdered graphene has also been combined with mesoscale aviation fuel (Huang, X.; Li, S. 2016). A mesoscale means that ignition and combustion tests simulated atmospheric conditions. Among the findings and conclusions, it is highlighted that the nanometric graphene in the fuel remains more stable than in micrometric size, therefore, the smaller size tends to improve the mixture. On the other hand "lA liquid jet fuel film containing graphene powders can be ignited by induction with a short delay time". It is also indicated that "graphene combustion predates ignition of vaporized fuel in reactors" and an important detail "graphene serves as a nucleation point to accelerate vaporization of aircraft fuel" given its adsorption capacity, already analyzed in the co2 uptake and the nucleation of ice crystals.
Opinions
The use of graphene oxide in aviation fuels could explain the presence of graphene oxide in the atmosphere, together with soot as a result of incomplete pyrolysis in aircraft reaction engines, as observed (Pöschl, U. 2005). In fact, all the scientific investigations consulted coincide in improving the performance and qualities of aviation fuel, when adding graphene oxide and derivatives additives. So,it can be said that the result of the combustion or pyrolysis of aviation fuel can generate traces of graphene oxide (in addition to soot) in the form of chemtrails together with water vapor obtained by condensation of exhaust gases from jet aircraft turbines from certain troposphere altitudes where the temperature is below the point of freezing. As explained in the entry on the nucleation of ice crystals in graphene oxide nanoparticles, it would not be negligible to think that graphene oxide in conditions of temperature and humidity such as those found at the flight height of commercial aircraft, caused the generation of clouds, which would explain cloud seeding and a climate modification or geoengineering effect. It would also explain the presence of graphene oxide in water droplets, as it has been warned. All this would confirm the existence of chemtrails as a result of the residue of the combustion of kerosene and graphene oxide in aviation jet engines (civil/military) and would involve evidence of air pollution.
On the other hand, the great frequency of appearance of GO graphene oxide has been observed, combined with Fe3O4. To the already known applications (CO2 adsorption, anticancer DNA vaccines, biocides-fertilizers for agricultural use, absorption of 5G electromagnetic waves ...) the application in aviation fuels is added. This means that GO / Fe3O4 or Fe3O4 / GO could be the universal compound from which the origin of all problems related to c0r0n @ v | rus is derived.
Anti-cancer DNA vaccines (Shah, M.A.A.; He, N.; Li, Z.; Ali, Z.; Zhang, L. 2014)
Biocides and fertilizers (Zhang, M .; Gao, B.; Chen, J.; Li, Y.; Creamer, A.E.; Chen, H. 2014)
Absorption of 5G electromagnetic waves (Ma, E.; Li, J.; Zhao, N.; Liu, E.; He, C.; Shi, C. 2013)
Vaccines with genetic reformulations CRISPR (bbott, T.R.; Dhamdhere, G.; Liu, Y.; Lin, X.; Goudy, L .; Zeng, L .; Qi, L.S. 2020)
Damage to graphene in the human body, see bibliographic repertoire and posts from this blog.
Finally, it remains to be determined to what extent direct graphene oxide fumigation would be feasible in order to reduce CO2 and collaborate in the fight against climate change. This point will be addressed in the next entry.
Bibliography
Askari, S.; Lotfi, R.; Rashidi, A.M .; Koolivand, H.; Koolivand-Salooki, M. (2016). Reological and thermophysical properties of ultra-stable kerosene-based graphene / Fe3O4 nanofluids for energy conservation = Rheological and thermophysical properties of ultra-stable kerosene-based Fe3O4/Graphene nanofluids for energy conservation. Energy Conversion and Management, 128, pp. 134-144. https://doi.org/10.1016/j.enconman.2016.09.037
Askari, S.; Rashidi, A.; Koolivand, H. (2019). Experimental research on the thermal performance of ultra-stable kerosene-based MWCNT and graphene nanofluids = Experimental investigation on the thermal performance of ultra-stable kerosene-based MWCNTs and Graphene nanofluids. International Communications in Heat and Mass Transfer, 108, 104334. https://doi.org/10.1016/j.icheatmasstransfer.2019.104334
Agarwal, D.K .; Vaidyanathan, A.; Kumar, S.S. (2016). Experimental research on the thermal performance of kerosene-graphene nanofluide = Experimental investigation on thermal performance of kerosene – grahene nanofluid. Experimental Thermal and Fluid Science, 71, pp. 126-137. https://doi.org/10.1016/j.expthermflusci.2015.10.028
Betiha, M.A.; Rabie, A.M .; Ahmed, H.S.; Abdelrahman, A.A.; El-Shahat, M.F. (2018). Oxidative desulfurization using graphene and its compounds for fuel containing thiophene and its derivatives: an update review = Oxidative desulfurization using graphene and its composites for fuel containing thiophene and its derivatives: An update review. Egyptian journal of petroleum, 27 (4), 715-730. https://doi.org/10.1016/j.ejpe.2017.10.006
Dai, Y.; Nie, G.; Gong, S.; Wang, L .; Pan, L .; Fang, Y.; Zou, J.J. (2020). Improved graphene oxide emulsification for synthesis in a single fuel container for high-density aircraft = Reduced graphene oxide enhanced emulsification for one-pot synthesis of high-density jet fuel. Fuel, 275, 117962. https://doi.org/10.1016/j.fuel.2020.117962
Feng, M .; Jiang, X.Z.; Mao, Q.; Luo, K.H.; Hellier, P. (2019). Initiation mechanisms for improved pyrolysis and oxidation of JP-10 (exo-tetrahydrodicyclopentadiene) in functionalized graphene sheets: knowledge of ReaxFF molecular dynamics simulations = Initiation mechanisms of enhanced pyrolysis and oxidation of JP-10 (exo-tetrahydrodicyclopentadiene) on functionalized graphene sheets: Insights from ReaxFF molecular dynamics simulations. Fuel, 254, 115643. https://doi.org/10.1016/j.fuel.2019.115643
Huang, X.; Li, S. (2016). Ignition and combustion characteristics of liquid aviation fuel film containing graphene to mesoscale powders = Ignition and combustion characteristics of jet fuel liquid film containing graphene powders at meso-scale. Fuel, 177, pp. 113-122. https://doi.org/10.1016/j.fuel.2016.03.004
Khaled, M. (2015). Adsorption performance of multi-walled carbon nanotubes and graphene oxide for the removal of thiophene and dibenzothiophene from model diesel fuel = Adsorption performance of multiwall carbon nanotubes and graphene oxide for removal of thiophene and dibenzothiophene from model diesel fuel. Research on Chemical Intermediates, 41 (12), pp. 9817-9833. http://dx.doi.org/10.1007%2Fs11164-015-1986-5
Pöschl, U. (2005). Atmospheric aerosols: composition, transformation, effects on climate and health = Atmospheric aerosols: composition, transformation, climate and health effects. Angewandte Chemie International Edition, 44 (46), pp. 7520-7540. https://doi.org/10.1002/anie.200501122
Rafique, I.; Kausar, A.; Anwar, Z.; Muhammad, B. (2016). Exploration of epoxy resins, hardening systems, and epoxy / carbon nanotube composite designed for high performance materials: A review. Polymer-Plastics Technology and Engineering, 55 (3), pp. 312-333. https://doi.org/10.1080/03602559.2015.1070874
Yadav, A.K .; Nandakumar, K .; Srivastava, A.; Chowdhury, A. (2019). Combustion of rocket grade kerosene drops loaded with graphene nanoplastics: a search for the reasons behind the optimal mass loads = Combustion of rocket-grade kerosene droplets loaded with graphene nanoplatelets — A search for reasons behind optimum mass loadings. Combustion and Flame, 203, pp. 1-13. https://doi.org/10.1016/j.combustflame.2019.01.030
Ignition and combustion characteristics of jet fuel liquid film containing graphene powders at meso-scale
https://www.sciencedirect.com/science/article/abs/pii/S0016236116300345?via%3Dihub
And to believe that 2016 was the end of that is a mistake!
Reduced graphene oxide enhanced emulsification for one-pot synthesis of high-density jet fuel
https://www.sciencedirect.com/science/article/abs/pii/S0016236120309583?via%3Dihub
Graphene oxide doped ethanol droplet combustion: Ignition delay and contribution of atomization to burning rate
https://www.sciencedirect.com/science/article/abs/pii/S0010218021004910
Influence of equivalence ratio on emissions in meso-scale vortex combustor
Ali Houssein Asmayou, Mazlan Abdul Wahid, Mohammed Bashir Abdulrahman, Mohsin Mohd Sies
2023, RECENT ADVANCEMENTS IN THERMOFLUID ENGINEERING - Proceeding
Advances in inorganic nanoparticles trapping stiffness measurement: A promising tool for energy and environmental study
Xiaolin Chen, Jiajie Chen, Jianxing Zhou, Xiaoqi Dai, Yuhang Peng, Yili Zhong, Ho-Pui Ho, Bruce Zhi Gao, Han Zhang, Junle Qu, Yonghong Shao
2023, Energy Reviews
Effect of Nanoparticle Addition on Evaporation of Jet Fuel Liquid Films and Nanoparticle Deposition Patterns during Evaporation
Linhui Ye, Shengji Li, Xuefeng Huang
2022, Langmuir
Understanding of Contradiction on Concentration Effect on Stability, Physical Properties, Evaporation and Microexplosion Characteristics of Al/JP-10/Oleic Acid Nanofluid Fuel
Qianmei Yang, Shengji Li, Linhui Ye, Xuefeng Huang
2022, Nanomaterials
Effect of Nanoparticle Concentration on Physical and Heat-Transfer Properties and Evaporation Characteristics of Graphite/n‑Decane Nanofluid Fuels
Shengji Li, Qianmei Yang, Linhui Ye, Hongzhe Du, Zhenzhong Zhang, Xuefeng Huang, Jiangrong Xu
2022, ACS Omega
Progress towards nanoengineered energetic materials
Richard A. Yetter
2021, Proceedings of the Combustion Institute - Article
Functionalized graphene sheet as a dispersible fuel additive for catalytic decomposition of methylcyclohexane
Hyung Sub Sim, Richard A. Yetter, Sungwook Hong, Adri C.T. van Duin, Daniel M. Dabbs, Ilhan A. Aksay
2020, Combustion and Flame
Combustion Properties of Fuels and Methods to Improve Them
Lun Pan, Xiu‐Tian‐Feng E
2020, High‐Energy‐Density Fuels for Advanced Propulsion - Chapter
Add to Library
Design and Synthesis of Nanofluid Fuels
Lun Pan, Xiu‐Tian‐Feng E, Jinwen Cao, Kang Xue
2020, High‐Energy‐Density Fuels for Advanced Propulsion
Tuning the agglomeration and combustion characteristics of aluminized propellants via a new functionalized fluoropolymer
Wen Ao, Peijin Liu, Huan Liu, Shixi Wu, Bowen Tao, Xuefeng Huang, Larry K.B. Li
2020, Chemical Engineering Journal
Dispersion Stability, Physical Properties, and Electrostatic Breakup of Surfactant-Loaded Aluminum/n‑Decane Nanofluid Fuel: Nanoparticle Size Effect
Shengji Li, Hongzhe Du, Zhu Zhuo, Yibin Lu, Xuefeng Huang
2019, Energy & Fuels
Initiation mechanisms of enhanced pyrolysis and oxidation of JP-10 (exo-tetrahydrodicyclopentadiene) on functionalized graphene sheets: Insights from ReaxFF molecular dynamics simulations
Muye Feng, Xi Zhuo Jiang, Qian Mao, Kai H. Luo, Paul Hellier
2019, Fuel
Micro-power generation using micro-turbine (moving) and thermophotovoltaic (non-moving) systems
Seyed Ehsan Hosseini
2019, Proceedings of the Institution of Mechanical Engineers Part A Journal of Power and Energy
Photocurrent enhancement on TiO2 nanotubes co-modified by N+ implantation and combustion of graphene
Jing Han, Xinggang Hou, Huanli Liu, Jing Li, Jianghong Yao, Dejun Li, Ping Wu
2019, Materials Letters
Reconstruction model for temperature and concentration profiles of soot and metal-oxide nanoparticles in a nanofluid fuel flame by using a CCD camera* * Project supported by the National Natural Science Foundation of China (Grant No. 51576100) and the Project of “Six Talent Summit” of Jiangsu Province, China (Grant No. 2014-XNY-002).
Guannan Liu, Dong Liu
2018, Chinese Physics B
Review on synthesis and properties of high-energy-density liquid fuels: Hydrocarbons, nanofluids and energetic ionic liquids
Xiangwen Zhang, Lun Pan, Li Wang, Ji-Jun Zou
2018, Chemical Engineering Science
Inverse radiation analysis for simultaneous reconstruction of temperature and volume fraction fields of soot and metal-oxide nanoparticles in a nanofluid fuel sooting flame
Guannan Liu, Dong Liu
2018, International Journal of Heat and Mass Transfer
Thermal performance of a meso-scale combustor with electrospray technique using liquid ethanol as fuel
Yunhua Gan, Xiaowen Chen, Yang Tong, Xia Zhang, Yang Zhang
2018, Applied Thermal Engineering
Droplet lasers: a review of current progress
D McGloin
2017, Reports on Progress in Physics - Article